Dr. Jeff Halter, Pediatric Surgeon at Maine Medical Center, likes to see his patients go home happy and healthy after surgery. Unfortunately, for many of his patients who use a gastrostomy tube, or g-tube, a return trip to the operating room is inevitable.
Gastrostomy tubes, or g-tubes, are feeding tubes placed through the abdomen directly into the stomach that provide patients with life-sustaining medications and nutrition. When the patient’s health condition improves, the g-tube is removed and the remaining wound typically heals within six weeks. But for up to 40 percent of patients, the wound never heals and forms a persistent tract called a gastrocutaneous fistula. After weeks of wound leakage, patient suffering and parent frustration, these pediatric patients are faced with additional, costly surgery.
Jeff believes that there is a better, safer way to remove g-tubes and encourage quicker healing. In 2019, he joined the Maine Medical Center Innovation Cohort with his early idea for a gastrocutaneous fistula closure device — an implantable clip that can be inserted through the g-tube site at the time of removal without surgery or anesthesia. The “Halter Clip” aims to seal the tract from inside the stomach while creating a chemical reaction in the tract that promotes safe healing. All of this can be done in one outpatient office visit.
Following the Innovation Cohort, Jeff filed a patent with the expertise and financial support of MaineHealth Innovation and was introduced to The University of Southern Maine engineering department who helped him create additional prototypes. In spring 2021, Jeff received $20,000 from the MaineHealth Innovation Ignite Fund and was introduced to Looma, LLC, in Saco, ME, to create his next prototype.

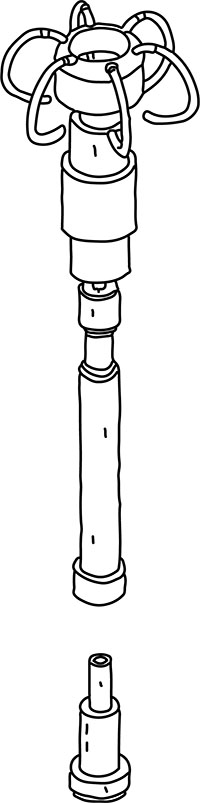
An early prototype of the Halter Clip innovation
With more than 500,000 g-tubes in use in the U.S., and millions worldwide, the Halter Clip has the potential to improve the quality of life for thousands of patients who suffer from g-tube complications. The Halter Clip is designed to be administered in an outpatient visit, decreasing the present demand on hospitals and
delivering value for both patients and care team members.
NEXT STEPS
Jeff plans to test the clinical grade prototype before pursuing FDA approval to conduct a small clinical trial. He also plans to apply for matching funds from Maine Technology Institute to further his product development.
TEAM
Jeff Halter, MD
Pediatric Surgeon
Maine Medical Center
Our Purpose
MaineHealth Innovation builds connections to drive diversity of thought, educates to produce creative problem-solvers and funds to accelerate ideas. By leveraging the ideas, insights and expertise of all care team members to develop novel solutions to our unmet care needs, we are working together so our communities are the healthiest in America.
